Clinicoserological factors associated with response to steroid treatment and recurrence in patients with IgG4-related ophthalmic disease. International Consensus Guidance Statement on the management and treatment of IgG4-related disease. 
Khosroshahi A, Wallace ZS, Crowe JL, Akamizu T, Azumi A, Carruthers MN, et al. 
The treatment outcomes in IgG4-related orbital disease: a systematic review of the literature. 2014 43:806–17.ĭetiger SE, Karim AF, Verdijk RM, van Hagen PM, van Laar JAM, Paridaens D. Ophthalmic manifestations of IgG4-related disease: single-center experience and literature review. IgG4-related disease in the eye and ocular adnexa. It rapidly reduces ocular inflammation and serum IgG4 levels to avoid excessive corticosteroid usage and reduce potential risk of adverse events. Obinutuzumab is a safe and promising therapeutic option for IgG4-ROD. Five patients (62.5%) experienced infusion-related reactions (IRRs) during the first obinutuzumab infusion, while only one (12.5%) experienced IRRs during all subsequent eight infusions. In patients with CR, the serum IgG4 levels at baseline correlated positively with dose numbers required for CR ( r = 0.86, P < 0.05). The serum IgG4 level correlated well with IgG4-RD RI at baseline and that after each treatment ( r = 0.852, P < 0.01 r = 0.78, P < 0.001). Six patients achieved complete remission (CR) (75%) and two patients achieved partial remission (25%). 
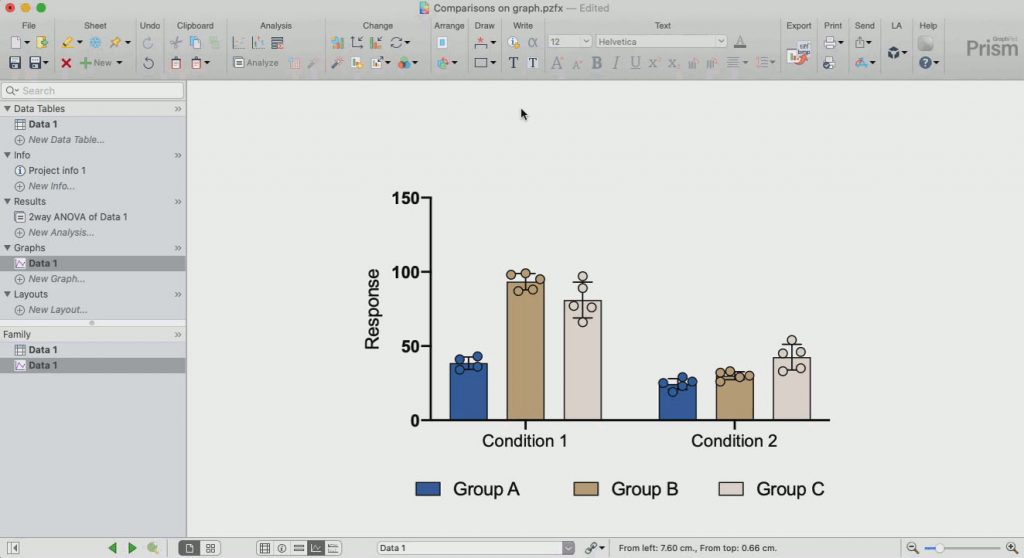
The mean IgG4-RD RI scores of all patients at baseline (7.75 ± 2.92) and after treatment (2.00 ± 0.76) were highly significantly different ( P < 0.001). The number of treatment sessions was based on treatment response. They were intravenously administered 1000 mg obinutuzumab at baseline and examined for changes in physical signs, orbital structure imaging parameters, IgG4-related disease responder index (IgG4-RD RI), serological index, and adverse events during treatment. MethodsĮight IgG4-ROD patients were retrospectively enrolled. We've fixed a bug in Friedman repeated-measures nonparametric one-way ANOVA that caused Prism to give incorrect results when some rows of data were excluded.You can nudge survival curves to prevent overlap.A new keyboard shortcut (Windows: Shift-Ctrl-V, Mac: Shift-Command-V) brings up the Paste Special dialog.A new keyboard shortcut (Windows: F9, Mac: Command-K) now recomputes simulated data (with new random error).The Output tab of the nonlinear regression dialog now lets you choose to test the residuals for normality.We've fixed a problem with the way Prism performs post tests following repeated-measures two-way ANOVA.Prism 4.03 (Windows) and 4.To evaluate the therapeutic efficacy and safety of obinutuzumab in remission induction for IgG4-related ophthalmic disease (IgG4-ROD) patients. Prism 4.02 and 4.01 (Windows) and Prism 4.0a (Macintosh) changes: After using Duplicate family of sheets, you can rename data table and the related sheets will rename too.Improved initial values in nonlinear regression of dose-response curves.Linear regression can now compute a prediction band when the line is forced through the origin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
